How High-Definition Spatial Proteomics Boosts Immuno-Oncology Therapy Development
High-definition spatial proteomics revolutionizes tissue imaging and analysis
Immuno-oncology (IO) has given clinicians incredible new opportunities for treating their patients, but it’s done even more than that. The focus on identifying and developing novel IO therapies — and the need to stratify the potential treatment population into responders and non-responders along the way — has also given pharma and biotech scientists the chance to dramatically improve how they characterize and validate new candidates.
Recently, mounting evidence has proven that the success of IO treatments depends just as much on the tumor microenvironment as it does on which proteins are expressed. This has ushered in remarkable advances in spatial biology — a new field designed to analyze the activity of genes or proteins in their native environment and architecture. As we now know, it matters a lot to cancer treatment outcomes not only which specific cell types are present, but how those cells are arranged, what kinds of cells surround them, and how those cells interact with one another.
We have never had this detailed a view of in situ biology before, and quite frankly, getting here hasn’t been easy. Scientists and engineers have had to design new technologies and new multiplexing approaches for collecting this information. Traditional techniques, such as immunohistochemistry (IHC), simply cannot provide the volume of information needed for such high-resolution interrogations of biological samples.
Spatial proteomic information with single-cell resolution
But while qualitative IHC can’t deliver enough data for a spatial biology approach, IHC remains the gold standard for analyzing samples and few scientists in pharma and biotech want to eliminate it. The ideal solution would be a quantitative version of IHC, an approach with the same trusted elements of this veteran technology that adds the ability to detect dozens of biomarkers at once.
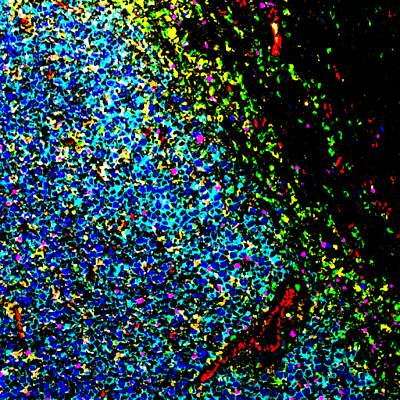
MIBI ENABLES DEEPER UNDERSTANDING OF THE TUMOR MICROENVIRONMENT
That’s where Ionpath comes in. Our team is pioneering high-definition spatial proteomics and revolutionizing tissue imaging and analysis to accelerate drug discovery and clinical study testing. Our MIBI™ technology, multiplexed ion beam imaging, is based on advances developed at Stanford University by world leaders in pathology and spatial biology. We deploy time-of-flight mass spec as a novel imaging platform to deliver high-definition spatial proteomics: spatial proteomic information, at single-cell resolution, with visualization and quantification of as many as 40 protein biomarkers in a single scan. It’s an incredible way to profile the architecture of the tumor microenvironment.
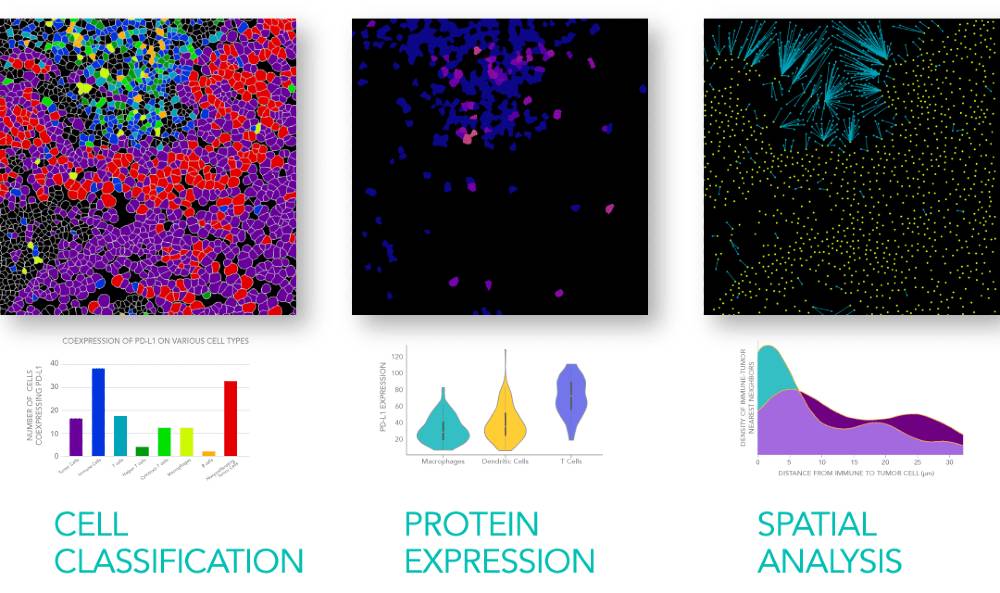
Meaningful data for translational research
With so many spatial biology tools becoming available, it can be difficult to figure out which one is right for your project or drug development program. Here’s how we look at it: researchers in pharma and biotech are eager for new and actionable insights, not mountains of potentially meaningless data. That’s why we also offer MIBI technology through a translational research service, allowing our team of pathology and computational experts to perform custom staining, imaging, and analysis of FFPE or other sample types. Scientists can opt for our pre-validated antibody panels for human or mouse tissue, and can add their own antibodies of interest that we validate for use with the MIBIscope™ platform. The end result: comprehensive enumeration of immune cell populations, quantification of checkpoint marker expression on a single-cell basis, and spatial context — all viewable in an easy-to-use, interactive, cloud-based data visualization interface.
While IO is the most obvious application for MIBI technology, we also work closely with researchers looking for insights in autoimmune disorders, infectious diseases, and neuroscience.
Interested in learning more? Request a pilot project or check out details about MIBI technology.